PG Diploma in Regulatory Affairs – Biologics, Vaccines & Biosimilars
Job Overview
-
Date PostedMarch 22, 2026
-
Location
-
Expiration dateApril 21, 2026
Job Description
PG Diploma in Regulatory Affairs – Biologics, Vaccines & Biosimilars
📌 Comprehensive Regulatory Strategy & CMC Training
Step into the rapidly growing biologics and vaccine regulatory domain with our specialized program designed for biotechnology, pharma, and life-science professionals.
🧬 Program Focus Areas
New Chapter starting on Drug Substance CMC Strategy
🔹 Upstream Process Development & Scale-Up
• Bioprocess development strategies for biologics and biosimilars
• Case studies on vaccines, therapeutic proteins & monoclonal antibodies (mAbs)
🔹 Downstream Process Development & Scale-Up
• Purification strategies & process optimization
• Case studies covering vaccines, biologics therapeutics & mAbs
🔹 CMC Data Preparation for Global Filings
• End-to-end preparation of CMC documentation for regulatory submission
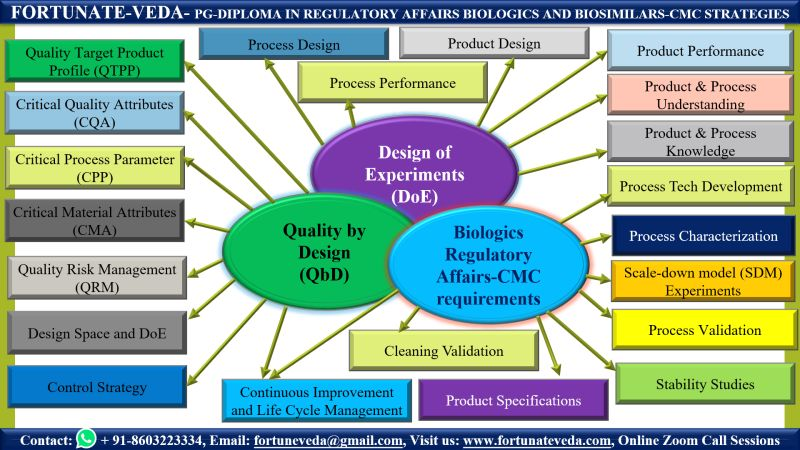
📊 Quality by Design (QbD) in Bioprocess R&D
• QbD principles in biologics process development
• QTPP (Quality Target Product Profile) preparation
• CQA, CPP & CMA identification strategies with case studies
• Quality Risk Management (QRM) approaches
• Design Space definition through process characterization
• Control Strategy development
• Continuous Process Verification & Lifecycle Management
📈 Biostatistical Tools Covered
• Minitab
• JMP
• ANOVA applications in bioprocess optimization
🧪 Advanced Regulatory Topics
• Cell & Gene Therapy – Regulatory CMC Strategy
• Process Technology Transfer (Drug Substance & Drug Product)
📅 New Batch Starting
🗓 23 March 2026
⏰ Time: 20:30 – 22:00 IST
💻 Mode: Live Online Sessions (Zoom)
📩 Contact & Registration
📧 Email: fortuneveda@gmail.com
🌐 Website: www.fortunateveda.com
📱 WhatsApp: +91-8603223334
👉 Enroll now to secure your seat in this specialized certification program.